By most accounts, prescription drug prices are often 50 to 60 percent higher in the United States than in other industrialized countries. These are frequently drugs that have been developed and manufactured in the U.S. yet, due to foreign price controls, are sold at bargain prices elsewhere. These price disparities have created incentives for the illegal importation of prescription drugs from abroad. In 2003, an estimated $695 million worth of drugs entered the U.S. from Canada alone.1 In addition, the Department of Health and Human Services’ (HHS) Task Force on Drug Importation estimates that an equivalent amount of drugs are being imported from the rest of the world each year.
Though the practice remains illegal, it has been facilitated by various state and local governments. These policymakers, pressured by local consumer groups, have adopted a shortsighted approach to deal with the problem of high brand name prescription drug prices. Following a similar approach, on February 9, 2005, Senator Chuck Grassley of Iowa introduced the Pharmaceutical Market Access and Drug Safety Act of 2005, a bill that would make it legal for U.S. consumers to buy prescription drugs from foreign sources. Included in the bill are provisions designed to prevent drug makers from limiting importation (by restricting sales to exporting pharmacies, for instance).
This bill comes two months after a major report released by the HHS Task Force on Drug Importation documented a multitude of cost and safety concerns related to commercial prescription drug importation. While the bill attempts to deal with the safety concerns, it does not address economic problems associated with drug importation. The HHS report confirmed what many economists and industry experts have long argued: that drug importation would diminish R&D incentives and eventually lead to fewer new drug therapies. Rather than address the real problem, i.e., price controls in other industrialized nations, U.S. policymakers have, in essence, chosen short-term savings in drug spending and put the long-term benefits of new therapies–not to mention patient safety–at risk.
Fewer R&D dollars spent and, consequently, fewer new drug therapies approved represent clear public health costs that should be weighed against the short-run savings in drug spending. This is an issue to be considered by policymakers throughout the nation, but another issue, the economic impact of drug importation, should also be analyzed. Research-intensive regions like California, Massachusetts and Maryland have a stake in the success of an industry that has been, and will continue to be, a powerful engine for economic growth.
Politicians need to be conscious of the longer-term, far-reaching harmful effects that their well-intentioned but misguided policies on drug importation may have on their local citizens, companies and economy.
The Impact of Drug Importation and Price Controls on the Maryland Economy
Research and Development in the Maryland Economy
Maryland has done an excellent job of fostering a competitive bioscience cluster. Building upon an exceptional academic research base, including leading academic research institutions such as John Hopkins University and the University of Maryland, along with the presence of the National Institutes of Health, the state has positioned itself as a national leader in the industry. In fact, Maryland outperforms every state but Massachusetts on the Milken Institute’s new Biopharmaceutical Innovation Index, a measure of the state’s research, financial and human capital infrastructure.
While the state’s strong biopharmaceutical cluster rests on its academic research base, it only ranks eighth in the Institute’s Biopharmaceutical Risk Capital Funding indicator, “a key indicator of commercialization activity for some new biopharmaceutical technologies and business concept innovation.” According to the report, “A state’s biopharmaceutical risk capital funding determines the success rate it will have converting basic and advanced research into commercially viable biopharmaceutical products and services.”2 This appears to have been borne out in reality. Many of Maryland’s life sciences firms have yet to bring a product to market, although many are moving products into final testing stages; this is a critical point in the development of the local biotechnology industry.3
Already, the biopharmaceutical industry plays an important role in the region’s economy. In 2003, the industry generated over 35,100 jobs.4 Of those, 10,710 people were employed within the industry, while the remaining 24,390 were attributed to the industry’s powerful multiplier effects.5 As the industry matures, its impact on the economy is expected to grow further–another 62 percent increase in employment over the next decade, according to the Milken Institute.6
The value of a thriving bioscience sector has been recognized by a growing number of states. Many have introduced initiatives to strengthen the local industry by providing early-state venture capital, research parks and laboratories. Yet while some public policy efforts have, on the one hand, sought to strengthen the industry, regulatory and legislative pressures continue to threaten it.
Importation and Price Controls
The argument is often made that drug companies could lower prices and continue to fund the current level of R&D spending. And critics point to the industry’s large profit margins as evidence. However, these critics misunderstand the relationship between R&D spending and prescription drug prices.
Biotechnology and pharmaceutical firms are lured into competition and, subsequently, high levels of R&D spending by the prospect of what economists call “monopoly profits”: profits that can only be realized by procuring patent rights for new drugs. While it may be necessary for companies to reinvest profits in R&D, they require the incentive to do so initially. Proposals that remove patent protections or limit the potential monopoly profits thus diminish R&D spending in two ways: First, they lower a firm’s cash flow and thereby limit funds available for R&D, and second, they diminish a firm’s expected rate of return on R&D and thus remove incentives to fund future research efforts.
Economic Impact on the Maryland Economy
In September 2004, a Beacon Hill Institute study released by the Institute for Policy Innovation (IPI) reported that a nationwide price control policy implemented at the federal level could result in a loss of $14.8 billion (in net present value terms) in industrial R&D spending over the first 12 years of price-control implementation. The authors further estimated that the failure rate of drugs entering clinical trials due to economic reasons would increase by approximately 70 percent, so that the annual average number of new drugs approved each year would fall from 31 to just nine.
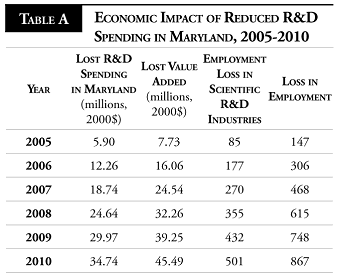
In Maryland, where the industry is on the brink of commercializing products, a drop in private R&D funding would be painful. Many biotechnology firms are very small and unprofitable. These innovators require capital inflows to fund further research. We estimate that in the first five years nearly $120 million in private R&D spending would be lost. Table A below illustrates the loss in R&D spending and the consequent economic impact of a price control policy.
Although there are varied methods of measuring economic impacts, the idea is straightforward. Initial spending in an economy has a “ripple” effect whose influence flows through to other sectors and households in the region. In essence, the initial spending in one sector brings about further spending in other sectors. This process creates new income and employment as it reverberates through the business community. Depending on the size of the initial impact, these ancillary effects can be quite large. For example, Boeing’s contribution to the Greater Seattle economy extends far beyond its initial outlay in wages and purchases.
In other words, expenditures have what economists call a “multiplier” effect that represents the recycling of money and income in an economy. By determining the multiplier for each category of expenditures, it is possible to simulate the initial spending and trace its influence through an economy. By measuring the change in economic indicators (employment, for instance) we can calculate the ultimate economic impact.
The economic contribution of R&D spending in Maryland consists of three types of impacts: direct, indirect and induced.
- The direct impact represents the economic impact directly attributable to the biotech firms: local purchases and employee compensation. For instance, in the pharmaceutical or biotech industries this may take the form of spending on legal services to secure patents. This spending creates income and employment directly for the industry’s vendors (legal services in this case).
- The indirect impact represents the spending done by other businesses supplying the goods and services demanded by the industry. For instance, the spending done by a local law firm as a result of being hired by a biotech firm creates employment and income for the law firms’ vendors.
- Finally, the induced impact refers to the income and employment created as a result of the spending done by the employees of the biotech industry, its intermediate suppliers and their vendors. Restaurants, real estate agents, gasoline stations, etc., all benefit from the local spending done by employees.
Using the IMPLAN model to describe commodity flows through the Maryland economy, we estimate the annual impact on the state economy as a result of the abandoned R&D spending.
We measure the economic impact using value-added as a measure of local economic activity; it represents the economic activity that ultimately stays in the Maryland economy. Included in value-added is employees’ wages, proprietors’ income, indirect business taxes and corporate profit.
The loss of R&D investment in Maryland has overarching effects on the state’s economy. The cumulative loss in employment for the period 2005-2010 is 867 jobs, many of these (501) in high-paying research positions. The lost R&D spending further results in a cumulative loss of $45.5 million in regional value-added.
While these figures are not large relative to the entire Maryland economy, it is significant in the nascent commercial bioscience industries which Maryland is taking such pains to promote. And all to implement an importation policy that may not provide the desired savings on drug spending. The HHS Task Force on Prescription Drug Importation recently reported that:
“Total savings to drug buyers from legalized commercial importation would be one to two percent of total drug spending and much less than international price comparisons might suggest. The savings going directly to individuals would be less than one percent of total spending. Most of the savings would likely go to third party payers, such as insurance companies and HMOs.”7
There thus remains the distinct possibility that policymakers could harm a vibrant local industry while failing to provide meaningful relief to consumers.
Conclusions
The biopharmaceutical industry has, and continues to be, a promising engine for economic growth in Maryland. The high earnings within the industry and powerful employment effects have, and will make it a valuable contributor to the region. Yet its future success remains vulnerable to regulatory and legislative pressures. As a home to some of the nation’s leading pharmaceutical firms, Maryland benefits from hundreds of millions of dollars in industrial R&D investment annually. In the process, thousands of high-paying jobs are created and new, innovative drugs are developed. Price control or importation policies designed to constrain prescription drug prices will, in the process, damper the incentive for the industry to engage in expensive and risky drug development. The result will be fewer new drug developments and fewer high-paying jobs in research-intensive states like Maryland. It is incumbent on policymakers to weigh these adverse effects against the desired, yet potentially elusive, savings that promoters of drug importation say it could provide.
Appendix: The IMPLAN Model
The IMPLAN economic impact modeling system is a product of Minnesota IMPLAN Group, Inc.
IMPLAN provides regional industry multipliers, which enable the user to provide detailed analyses of the direct, indirect and induced economic impacts on the local economy of a change in final demand for certain industries.
IMPLAN multipliers are designed to model a variety of scenarios and are traditionally used to model a shock to a regional economy. Examples of uses of the model include opening or closing military bases, new energy facilities, new sports stadiums, opening or closing manufacturing plants and airport or port facilities. All these scenarios are modeled by estimating changes in final demand by industry and entering them into the IMPLAN model for the region.
Any systematic analysis of economic impacts must account for the inter-industry relationships within a region. IMPLAN, accounts for inter-industry relationships through the use of a regional transaction table that is algebraically manipulated to produce a set of regional multipliers.
IMPLAN captures the direct effects of changes in final demand and local purchases made by local companies as a result of this increase in final demand. Because IMPLAN is based on regional industry multipliers it will also capture the ancillary effects arising from the income earned from the local companies’ input purchases.
IMPLAN is based on a national transaction table that is regionally adjusted through the use of Regional Purchase Coefficients (RPC). RPCs represent the portion of local demand purchased from local producers. Once the transaction table is regionalized, a coefficient matrix is derived by dividing each industry column by the column total. This coefficient matrix is also called the A matrix. Through the algebraic manipulation performed below the regional multipliers are derived:
X = (I -A)-1 Y ,
Where
X = Industry output,
I = Identity matrix,
A = A matrix,
Y = Final Demand.
This analysis accounts for changes in Y, in the form of R&D spending. For the purposes of this study, the IMPLAN model is used to determine how the loss in R&D spending translates into value added and employment losses throughout the economy.
Endnotes
- Health and Human Services Task Force on Drug Importation, “Report on Prescription Drug Importation,” Department of Health and Human Services, December 2004, page 11.
- Ross DeVol, Perry Wong, Armen Bedroussian, Lorna Wallace, Junghoon Ki, Daniela Murphy and Rob Koeppin, “Biopharmaceutical Industry Contributions to State and U.S. Economies,” Milken Institute, October 2004, page 111.
- William Patalon III, “Critical Stage for Biotechs,” The Baltimore Sun, January 7, 2005, page 1D.
- De Vol et al, October 2004, page 7.
- Ibid, page 68.
- Ibid, page 68.
- Health and Human Services Task Force on Drug Importation, “Report on Prescription Drug Importation,” Department of Health and Human Services, December 2004, page 65.
About the Institute for Policy Innovation
The Institute for Policy Innovation (IPI) is a nonprofit, non-partisan educational organization founded in 1987. IPI’s purposes are to conduct research, aid development, and widely promote innovative and nonpartisan solutions to today’s public policy problems. IPI is a public foundation, and is supported wholly by contributions from individuals, businesses, and other non-profit foundations. IPI neither solicits nor accepts contributions from any government agency.
IPI’s focus is on developing new approaches to governing that harness the strengths of individual choice, limited government, and free markets. IPI emphasizes getting its studies into the hands of the press and policy makers so that the ideas they contain can be applied to the challenges facing us today.
This study was prepared for the Institute for Policy Innovation by the Beacon Hill Institute at Suffolk University, Boston, MA.
